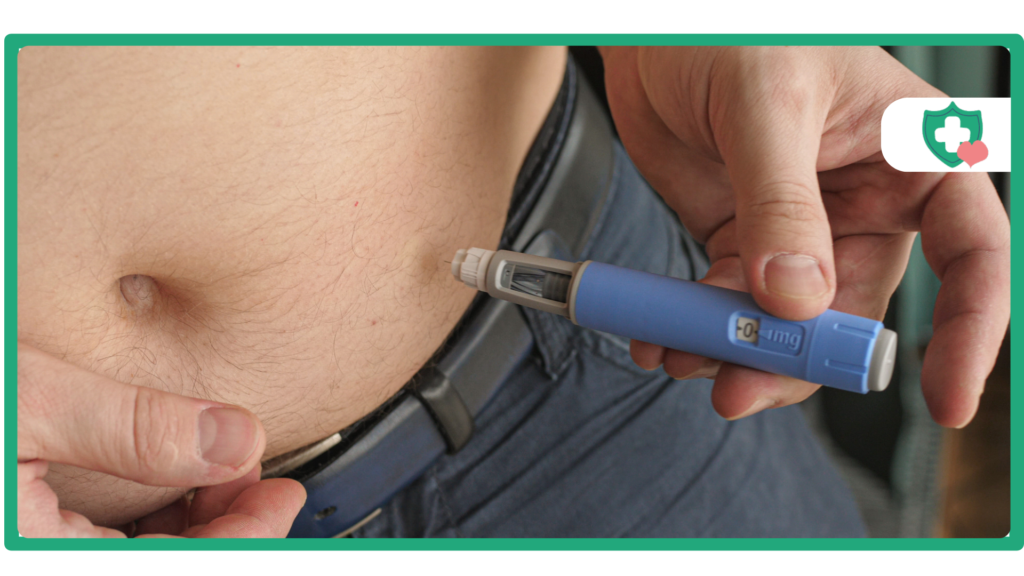
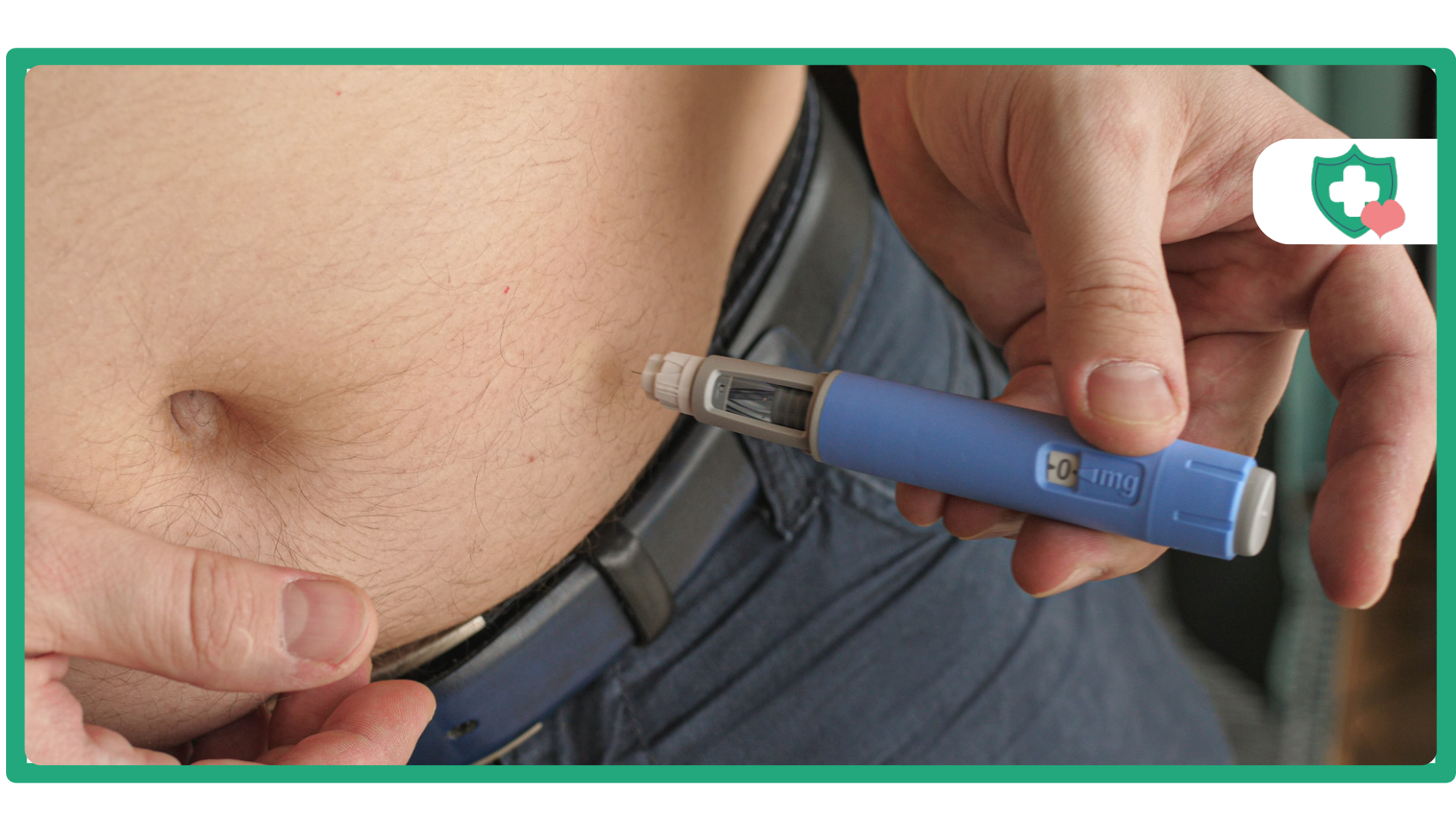
GLP-1 and GIP analogue therapies like Tirzepatide (which can be found under the brand names Mounjaro or Zepbound) are changing the way that providers manage people living with diabetes and the complicated world of obesity treatment. Unfortunately, some individuals may want to modify their dosage schedule (aka split dosing) because they think that receiving “more” or that “a different approach will work better” would provide them with better results. For those considering split dosing, there are important scientific, safety, and regulatory factors that you should be aware of when using this class of medications.
What Does Split Dosing Tirzepatide Mean?
Split dosing is the separation of a single dose recommended by the manufacturer into two or more smaller doses that can be given over the course of the week.
The Concept
Generally, a single weekly injection of Tirzepatide is given every week in standard Tirzepatide dosing protocols, whereas in split dosing, you would take that weekly volume and divide it into smaller volumes.
Example Scenario
If a patient is taking a weekly 10 mg dose, instead of taking all of that 10 mg at once (for example, on Sunday morning), it can be taken as a “Split Dose” as:
- 5 mg injected on Monday
- 5 mg injected on Thursday
The total amount of medication entering the body over seven days remains the same, but the delivery frequency is doubled.
Why Patients Consider It
Some patients consider this option, but their motivation is not to be “difficult,” but rather to enhance their experience. The reasons for doing this typically fall into three categories:
- Softening Side Effects: Tirzepatide is known to produce gastrointestinal side effects such as nausea (up to 28% for higher doses) and/or bloating. Many patients believe that doing smaller “doses” throughout the week would lessen the intensity of these side effects.
- Smoother Effects: Some users feel there is a “crash” effect (around days 5 or 6 of the week) due to declining plasma drug levels toward the end of the dosing interval and would like to find ways to keep the levels of appetite suppression more stable by administering their doses more frequently.
- Aligning with Cycles: Some patients try to schedule their doses for certain days of the week to avoid cravings or triggers they will encounter during the weekend or at other events related to their lifestyle.
Why FDA-Approved Tirzepatide Pens Cannot Be Split
While the logic of split dosing might seem sound on paper, the physical reality of the medication’s delivery system makes it not only difficult but dangerous for the average user.
Design and Packaging: Pre-filled, Single-Use Pens
Tirzepatide products generally prescribed include Mounjaro and Zepbound, which are dispensed through advanced auto-injector pens designed for single-dose delivery systems.
When activated by a push-button mechanism, an internal spring inside the pen releases the entire amount of liquid at once without any “half-click” or pause mechanisms. Once the needle is activated, the pen is empty. As a result, it is impossible to halt an injection after it has started, and because injecting via this method marks the end of the medicated pen’s life (it cannot be reused), all attempts to remove liquid from the pen will compromise its sterility.
Sterility and Stability Risks
Perhaps the most critical reason to avoid splitting is the preservative-free formulation.
- Contamination: Tirzepatide pens are manufactured in a sterile environment. They do not contain antimicrobial preservatives because they are meant to be used instantly.
- The Risk: If a patient attempts to “decant” the medication into a separate vial or syringe, they expose the liquid to air and bacteria. Without preservatives, that liquid becomes a breeding ground for pathogens, which can lead to serious skin infections or systemic illness upon the second injection.
Manufacturer and Regulatory Guidance
Eli Lilly and the FDA have provided explicit guidelines on the use of their product.
- No Manipulation: Pens and single-dose containers are intended for one-time use only. Do not reuse or alter the original product and discard any liquid left in the pen or vial.
- Discard Policy: If a device malfunctions and gives you fluid that does not belong in the vial used after the injection, throw away the fluid.
- Lack of Clinical Data: There is currently no clinical evidence to support that administering Tirzepatide in split doses has similar and lasting metabolic benefits or a long-term safety profile compared to once weekly subcutaneous administration.
Proper Medical Supervision for Dose Adjustments
When facing challenges related to medication, it is essential to seek professional help rather than attempting to create your own remedies at home. Clinical consultation with a healthcare professional is necessary as you navigate this type of treatment.
Consult Your Healthcare Provider
Your healthcare provider is the best person to guide and support you on your journey with Tirzepatide. If you’re experiencing extreme nausea and feeling weighed down right now, your healthcare provider needs to know so they may either prescribe medications to alleviate your symptoms or modify the way the medication is administered through a change in dosage levels over time (i.e., titration).
The Standard Titration Approach
For Tirezapride dosing, your clinician will begin with a low dose (2.5mg) for four weeks to allow your body to acclimate before increasing the dosage stepwise to the next level (5mg). If you are experiencing unpleasant side effects while titrating, your clinician can choose to give you another month on the lower dose instead of progressing to a higher dose early.
Monitoring and Safety
While you are taking Tirezapride, your clinician will monitor your titration rate based on the following three factors: glucose levels, kidney function, and overall health. By self-titrating or taking a higher-than-prescribed dose of Tirezapride, you are putting yourself at risk by eliminating the safety measures (regular monitoring) provided by your healthcare provider.
When Might Modified Dosing Be Considered?
There are some very specific clinical circumstances in which a physician may use a “modified” type of intervention (e.g., medicine or device) for an individual patient. There is a distinction between self-splitting and a clinical adaptation process.
- Extreme Sensitivity: A physician may keep a patient at a low dose (5mg) of a drug for an extended period if the patient was intolerant to increasing from 5 mg to 7.5 mg.
- Initial Adaptation Phase: Some patients who have had prior episodes of severe gastrointestinal (GI) problems may require their physician to prescribe their injectable medication on a different day of each week than other patients so that the peak toxicity occurs while they are at home and can rest.
- Unique Medical Circumstances: Many patients with unusual metabolic conditions require their schedule supervised and adjusted by an endocrinologist.
Proper Tirzepatide Administration Techniques
To achieve optimal results with your medication, pay attention to administering it correctly according to the official instructions rather than changing the amount you take (i.e., dosages).
Storage and Handling
- Refrigeration: Keep your pens cold (between 2-8°C or 36-46°F) by placing them in the fridge when not in use. If they are kept out of the fridge, they may remain stable for 21 days (up to 30°C or 86°F).
- Light Protection: Always store your pens in the original packaging to shield them from UV exposure.
- No Re-entry: Do not attempt to reuse any needles or vials from previously-used containers on new ones.
Injection Procedure
- Site Rotation: The injection sites available to you are your stomach, thigh, and back of your upper arm, which can be rotated with each use. If you use the same injection site every week, this will likely create ‘lumps’ under the skin or irritation at the injection site.
- Hygiene: An alcohol swab will need to be used to clean the injection site, and always wash your hands before giving yourself the injection.
- Depth: The auto-injector is designed to inject into the subcutaneous area, so you need to always ensure that the pen is held flat against the skin prior to unlocking and pushing down on the button.
Adherence to Schedule
Because Tirzepatide has a half-life ranging from 5 days to 1 week, it allows for weekly dosing. This means that, due to the half-life, the patient still has the drug in their system from prior doses at the time of their next dose. The “Steady State” refers to the amount of medication that remains in the body at a constant concentration after multiple doses have been taken. This steady state is considered to be reached when a 7-day or more gap between doses is maintained.
Conclusion
The desire to “hack” your medication schedule is a common response to the side effects of Tirzepatide; however, the dangers associated with splitting doses far outweigh any potential advantages that could be gained through this practice.
- Safety First: Tirzepatide pens are medical devices that were specifically manufactured to be used once as directed. Additionally, when using your pen for split doses, you are increasing the risk of infection and instability in the chemical composition of your medication.
- Follow the Science: The only evidence supporting the use of weekly doses of Tirzepatide as an effective and safe treatment option comes from the results of large clinical trials.
- Talk to Your Doctor: Your physician has several options available for modifying your plan safely. Do not attempt to change your dosage unless under the care of a qualified professional.
Common Questions About Tirzepatide Dosing
Is it permissible to split a tirzepatide pen dose?
No. The prefilled pens supplied by the manufacturers are mechanical, single-use devices and are not intended to be refilled once they have been used, nor can they be stopped during an injection, as they do not have any preservatives, so that once used, the medicines cannot be stored for any length of time after breaking the seal on the medication.
Is split dosing safe or effective?
There is no clinical evidence to confirm that split dosing is effective for weight loss or glucose control. From a safety standpoint, it is considered high-risk due to the potential for bacterial contamination and inconsistent dosing (as it is nearly impossible to measure exactly half a dose from a broken pen).
What action should be taken if side effects make split dosing seem necessary
If you have side effects leading you to consider splitting your dosage, please reach out to your healthcare professional as soon as possible. Your healthcare provider may provide you with treatment options such as dietary modifications (lowering fat intake often results in decreased nausea), antiemetic medications, or an adjustment to the titration schedule.
Resources
https://pi.lilly.com/us/mounjaro-us-ifu.pdf
https://pi.lilly.com/us/zepbound-us-ifu.pdf
https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/217806s000lbl.pdf
https://www.nejm.org/doi/full/10.1056/NEJMoa2206038
https://www.nejm.org/doi/full/10.1056/NEJMoa2107519
https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=15f75a5e-93e8-4bcc-8f8b-389db0b2fdbf
https://www.lilly.com/news/media/media-kits/mounjaro
https://www.fda.gov/media/169110/download