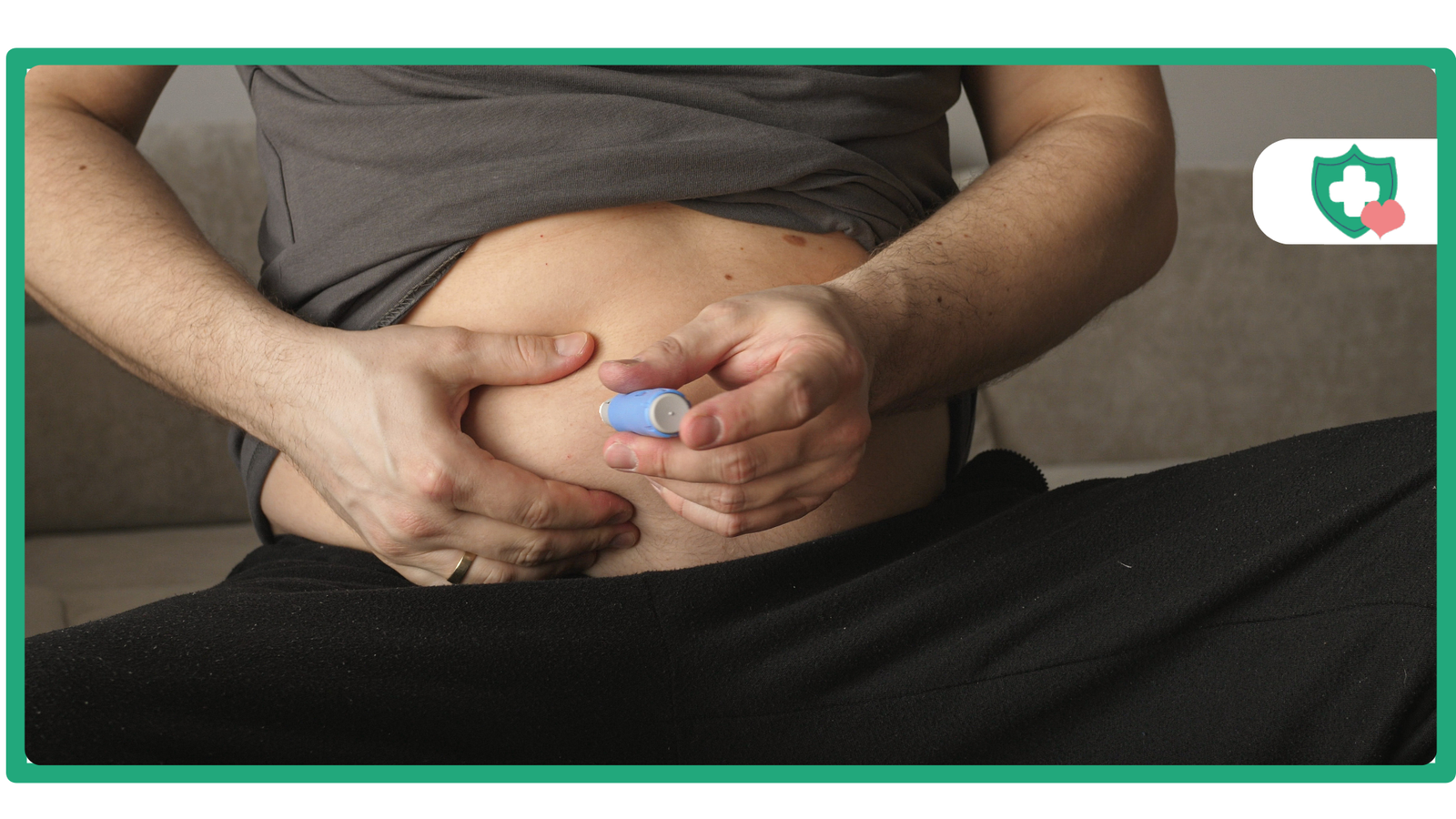
Advances in incretin mimetics have changed how we manage metabolic health and weight over the past few years. Tirzepatide is a leading example of this revolution, as a result of its ability to effectively promote weight loss and regulate blood sugar levels, surpassing many prior options.
As an increasing number of individuals consider using this treatment, one of the primary questions arising is, “Is it possible to use a pill instead of receiving a needle?” Although many patients may be interested in taking an oral form of the drug daily rather than using an injectable formulation, the current management of this product by the FDA through existing medical and biological knowledge makes the situation seem much more complicated when viewed from the perspective of medicine and pharmacology. This document examines the differences between oral and injected tirzepatide so as to help you make a decision about which is best suited for you.
Understanding Tirzepatide and Its Growing Popularity
Tirzepatide represents a new, “next-generation” treatment for obesity and Type 2 diabetes. It is different from previous medications designed to treat these conditions because most prior drugs targeted only a single hormonal pathway. Tirzepatide targets both the glucose-dependent insulinotropic polypeptide (GIP) hormone pathway and the glucagon-like peptide-1 (GLP-1) hormone pathway; this means that tirzepatide has a “double-barreled” impact on metabolism as a dual GIP/GLP-1 receptor agonist rather than just one.
The Clinical Breakthrough
The rapid rise in demand for tirzepatide has been driven by its effectiveness in clinical studies, where participants lost large percentages of their body weight, similar to patients who undergo gastric bypass surgery. In addition to promoting weight loss, it also provides significant benefits for overall metabolic health, including improved cholesterol levels and enhanced blood sugar control.
The Evolution of Administration
Because of the increasing demand for tirzepatide, there has been growing interest in finding different ways to deliver the medication. Some patients are afraid of needles, and others find it difficult to store and discard their pen devices. There is intense debate about whether an oral version would be as effective as an injection.
What Is Tirzepatide?
To choose the right format, you first need to understand what this molecule actually does inside your body.
How Tirzepatide Works
Tirzepatide acts on two primary receptors in the body to regulate how you process energy:
- GLP-1 Action: The slowing of gastric emptying from GLP-1 causes food to remain within the stomach for an extended period. As a result, consumers will experience feelings of satiety much sooner than with previous methods of food intake. In addition, GLP-1 agonists send signals to the brain’s appetite centers, thereby causing a decrease in food cravings.
- GIP Action: GIP is often referred to as the “secret sauce” in the efficacy of tirzepatide. It is believed that GIP is responsible for regulating fat metabolism through influencing the body’s metabolic rate. Furthermore, GIP may aid in decreasing the incidence of nausea associated with certain GLP-1 medication protocols.
FDA-Approved Medications
Currently, tirzepatide is marketed under two brand names, both manufactured by Eli Lilly:
- Mounjaro: Approved specifically for the treatment of Type 2 diabetes.
- Zepbound: Approved for chronic weight management in individuals with obesity or overweight status with at least one weight-related condition.
Oral Tirzepatide Explained
There is much anticipation for an eventual approval of oral tirzepatide by the FDA; however, it is also essential to have expectations aligned with what actually exists today.
Is Oral Tirzepatide FDA Approved?
The answer to this question is a definitive no; no oral formulation of tirzepatide has been approved by the FDA for commercial distribution as of January 2026. The only oral forms of tirzepatide available online are either being investigated in clinical trials or – even worse – are being marketed by rogue pharmacy operations that do not provide appropriate safety documentation.
Why Creating a Pill Is Difficult
- Peptide Breakdown: Since tirzepatide is a peptide (i.e., a chain of amino acids), it is broken down by your stomach into energy (fuel). When you ingest a peptide, the acid and enzymes in your stomach will usually destroy the peptide before it can be absorbed into your bloodstream.
- Poor Absorption: The lining of the gastrointestinal tract isn’t particularly adept at absorbing large molecules like tirzepatide, even if a peptide survives the acidic environment of the (GI) tract.
- Bioavailability: To achieve similar effects to those achieved with small injections, an oral dose of tirzepatide would have to be enormous, resulting in variable levels of the drug in your bloodstream.
Injectable Tirzepatide Overview
The injectable form is the “gold standard” mainly because it does not go through the digestive tract and delivers the medication directly into the subcutaneous fat for establishing a stable and consistent absorption into the bloodstream.
Administration and Dosing
Although tirzepatide (injected) is a needle-based therapy, it is easy to use (i.e., user-friendly). A prefilled, single-use pen is used to inject tirzepatide once a week. Patients typically report feeling minimal to no pain due to the use of a smaller needle diameter (typically a 29 – 34 gauge needle).
Clinical Research
- Dosing Titration: Starting low (2.5 mg) and increasing gradually to minimize side effects.
- Safety: Long-term monitoring for rare but serious risks.
- Efficacy: Predictable weight loss trajectories.
Oral vs. Injectable Tirzepatide: Key Differences
| Feature | Oral Tirzepatide (Experimental) | Injectable Tirzepatide (Approved) |
|---|---|---|
| FDA Approval | Not approved | Fully approved (Mounjaro/Zepbound) |
| Administration | Daily pill (Theoretical) | Once-weekly injection |
| Clinical Evidence | Limited/Investigational | Extensive/Robust |
| Absorption | Highly variable | Reliable and consistent |
| Weight Loss Results | Unproven in the general public | Clinically proven (up to 20%+) |
| Safety Profile | Unclear/Potential GI distress | Well-established |
Effectiveness and Expected Results
When choosing a medical treatment, effectiveness is usually the priority.
Weight Loss Outcomes
Injectable tirzepatide has been proven to produce superior results, as evidenced by a study showing most patients lose 15% to 22% of their body weight within 72 weeks from the time they start using the drug. Injectable tirzepatide is a sustained-release product that progressively releases a steady dose of the drug. This will create a consistent “fullness” or satiety effect throughout the week.
On the other hand, oral products have the issue of the “absorption ceiling.” With oral formulations, a large portion of the dose is lost during the digestive process, and, therefore, there isn’t enough of the active ingredient in the blood to provide a therapeutic effect for inducing substantial weight loss (without causing serious gastrointestinal distress).
Blood Sugar and Metabolic Benefits
For those with Type 2 diabetes, the injectable form has shown an incredible ability to lower A1C levels, often bringing patients into the “normal” range. Without peer-reviewed data on oral tirzepatide, doctors cannot guarantee similar metabolic protection with a pill.
Side Effects and Safety Considerations
All medications have side effects, but the delivery method changes how those risks manifest.
Common Side Effects (Injectable)
Most side effects of injectable tirzepatide are gastrointestinal and occur when doses are increased:
- Nausea and Vomiting
- Diarrhea or Constipation
- Indigestion or stomach pain
- Fatigue
Safety Concerns (Oral)
There are several risk factors associated with the illegal use of oral tirzepatide that are greater than any possible benefits. Because these drugs do not undergo any FDA regulation, therefore:
- Unknown Purity: The user has no way to know what exactly is contained within the ‘pill’ they are ingesting.
- Dosage Chaos: Because the manufacturer has no oversight, one pill may contain 10% of the drug while another pill contains 200% of the same drug, and this will cause extremely harmful fluctuations in blood levels.
- Long-term Unknowns: We do not know if long-term, daily exposure to tirzepatide (taken orally) is harmful to the esophagus and/or stomach lining after several years.
Which Option May Be Right for You?
Choosing a medication is a deeply personal decision made in consultation with a healthcare provider.
Injectable Tirzepatide Is Best For:
- Proven Results: If your goal is significant, life-changing weight loss backed by science.
- Convenience: If you prefer “setting and forgetting” your treatment once a week rather than remembering a daily pill.
- Safety: If you want a medication that has been through the rigors of FDA testing.
Oral Alternatives to Consider
Individuals who are completely averse to needles should not attempt to find illicit or “bootleg” oral tirzepatide. Instead, people should speak with their doctor about Rybelsus (oral semaglutide). While Rybelsus (oral semaglutide) has a different active ingredient than injectable tirzepatide (it only targets GLP-1), at this time, it is the only orally-administered peptide available by prescription that is approved by the FDA. Rybelsus does not possess as much potency compared to injectable tirzepatide; however, it is considered a safe and regulated option.
Cost and Accessibility
- Insurance: Most insurance companies will only cover FDA-approved medications. This means Mounjaro or Zepbound (injectables) may be covered, whereas an experimental or compounded oral version will almost certainly be an out-of-pocket expense.
- Accessibility: Because injectable tirzepatide is the only legal version, it is the only one you can reliably get from a primary care physician or a legitimate pharmacy.
Final Comparison Summary
Currently, injectable Tirzepatide is more effective than the oral version in treating obesity and type-2 diabetes. The injectable form is safe and effective through extensive clinical trials. The hope that tirzepatide will one day be available as a pill does exist, but until then, using unapproved oral forms could pose a risk to your well-being as they’re unlikely to work effectively.
For those with obesity and metabolic syndrome problems, the most effective first step is to speak with a doctor or metabolic specialist about using injectable tirzepatide and developing an appropriate plan based on individual needs.
Frequently Asked Questions
Is oral tirzepatide available as a pill?
No. While research is ongoing, there is currently no FDA-approved version of tirzepatide in pill form. Only the injectable versions (Mounjaro and Zepbound) are authorized for use.
Which is more effective for weight loss?
The injectable form is significantly more effective. Clinical trials have proven their results, whereas oral versions currently lack the data to support similar outcomes.
Are oral versions safe to use?
Currently, no. Since no oral version has passed FDA safety trials, the long-term risks to the digestive system and overall health remain unknown.
How often is injectable tirzepatide taken?
Weekly treatment through subcutaneous injection is typically performed on the same day each week. Its long-lasting injectability means it is administered just once every 7 days to avoid fluctuations in blood sugar levels and appetite due to changes in hormone levels.
Resources
- https://www.lilly.com/news/press-releases/lillys-zepbound-tirzepatide-delivered-up-to-20-percent-weight-loss-adults-obesity-or-overweight-ibm-two-clinical-studies
- https://www.mounjaro.com/hcp/clinical-trials
- https://www.nejm.org/doi/full/10.1056/NEJMoa2206038
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215866s000lbl.pdf
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/217806s003lbl.pdf
- https://www.lilly.com/discovery/pipeline/tirzepatide https://investor.lilly.com/news-releases/news-release-details/lilly-initiates-phase-3-clinical-trial-evaluating-oral-formulation
- https://www.nature.com/articles/s41573-020-0087-2
- https://www.rybelsus.com/
- https://www.mounjaro.com/safety-and-side-effects https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-patients-risks-associated-compounded-tirzepatide
- https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2802363
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215866s039lbl.pdf